Product Name
1.1 Brand Name
1.2 Generic Name
1.3 Full Product Name
Product name is defined as a name given to a product which may either be a proprietary name (an invented name); or a generic name (common name) or scientific name, together with a trade mark or the name of the manufacturer.
- Product name shall consist of dosage form and strength (for single active ingredient product). (e.g. XYZ Capsule 500mg)
- Any product name which is the same or similar either in writing/ pronunciation, with the product name of an adulterated product is prohibited.
- Product name may be included together with the brand name or trademark name, if applicable.
- Please check product names which are not permitted to be registered as specified in DRGD.
Reference National Pharmacy Regulatory Agency (formally known as National Pharmaceutical Control Bureau):
Drug Registration Guidance Document (DRGD) – Appendix 9 under 9.1.4 Table 2 List of Non-Permissible Product Names.
Health Supplements:
- Dosage form and strength of product would need to be entered as part of product name to allow for multiple dosage forms (e.g. tablet, capsule) and strengths (e.g. 200mg and 400mg) for any particular named (proprietary or generic) product.
- In any event if found that registered product name is similar to another registered product, NPRA reserve the rights to request for the change in the product name.
- Product with more than 1 active ingredient could not include strength of active ingredients in the product name.
Reference National Pharmacy Regulatory Agency (formally known as National Pharmaceutical Control Bureau):
Drug Registration Guidance Document (DRGD) – Appendix 4 under 4.5 Table 5 List of Non-Permissible Product Name for Health Supplements.
How to Access Product Name in QUEST System ?
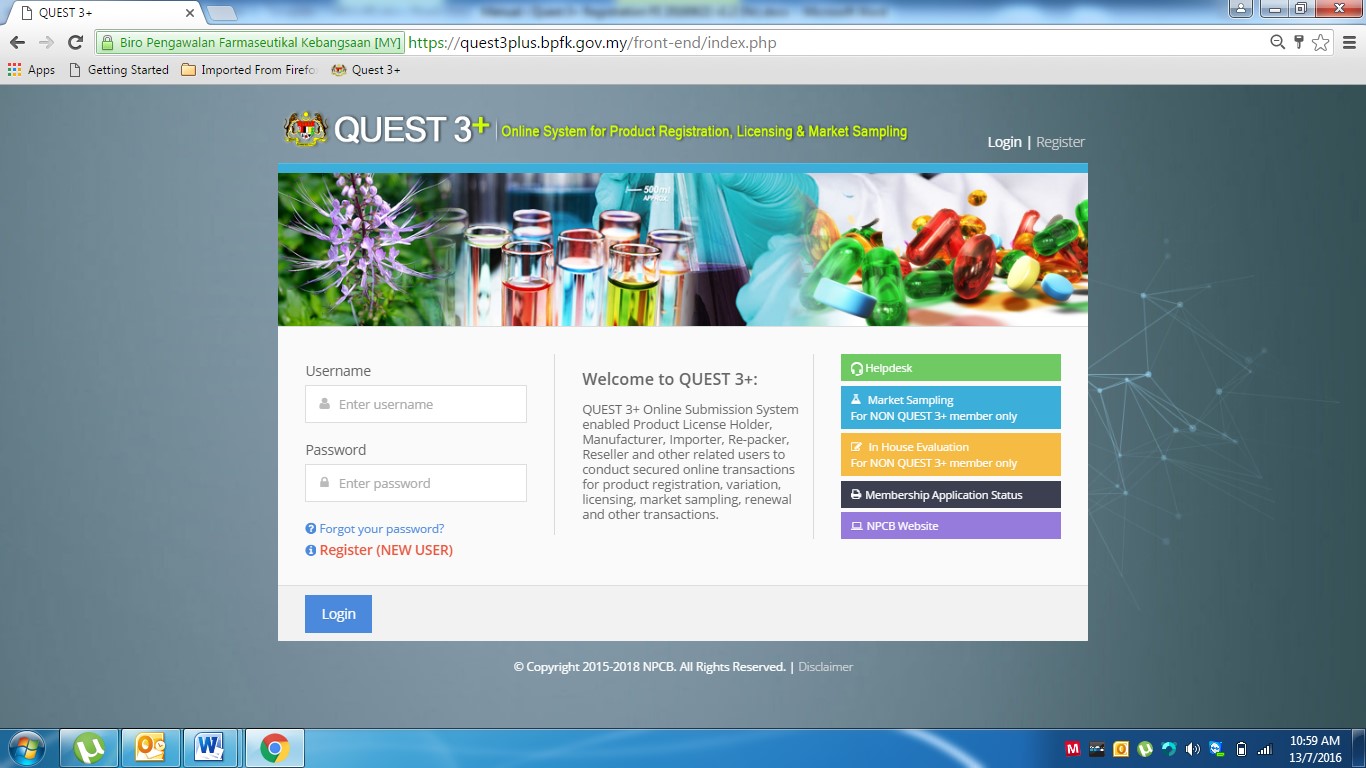
To access the Quest 3+ system, user need to use the URL as below:
https://quest3plus.bpfk.gov.my/front-end
Figure 1 as the above will appear and the user will need to enter the following information:
1. User Name: User ID
2. Password: Enter Password.
3. Click Login
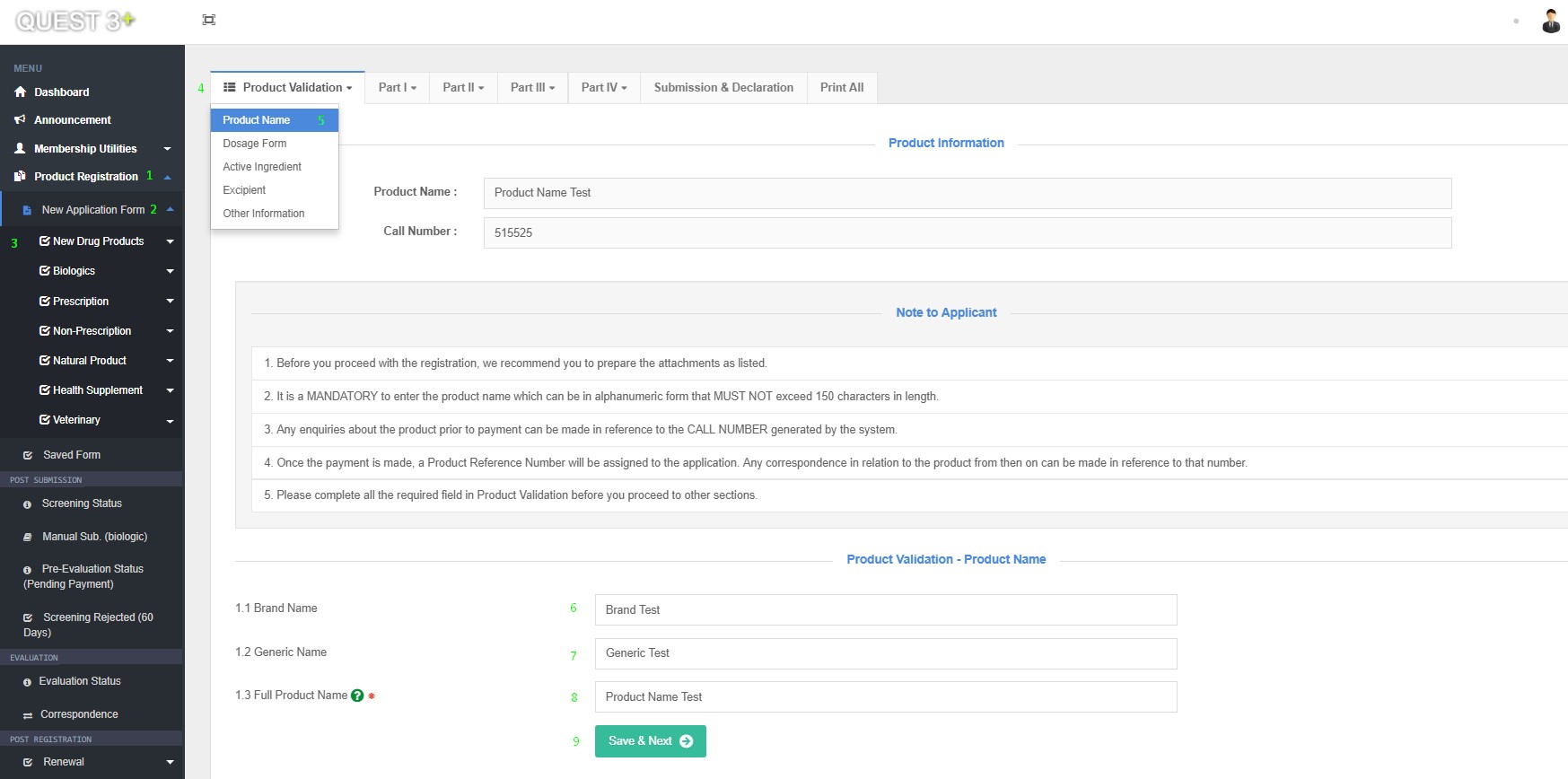
After successful log in, Submission form can be access here : Product Registration >> New Application Form >> Choose Product Category >> Product Validation >> Product Name
Fill in this Fields and click 'Save & Next' button :
1.1 Brand Name
1.2 Generic Name
1.3 Full Product Name